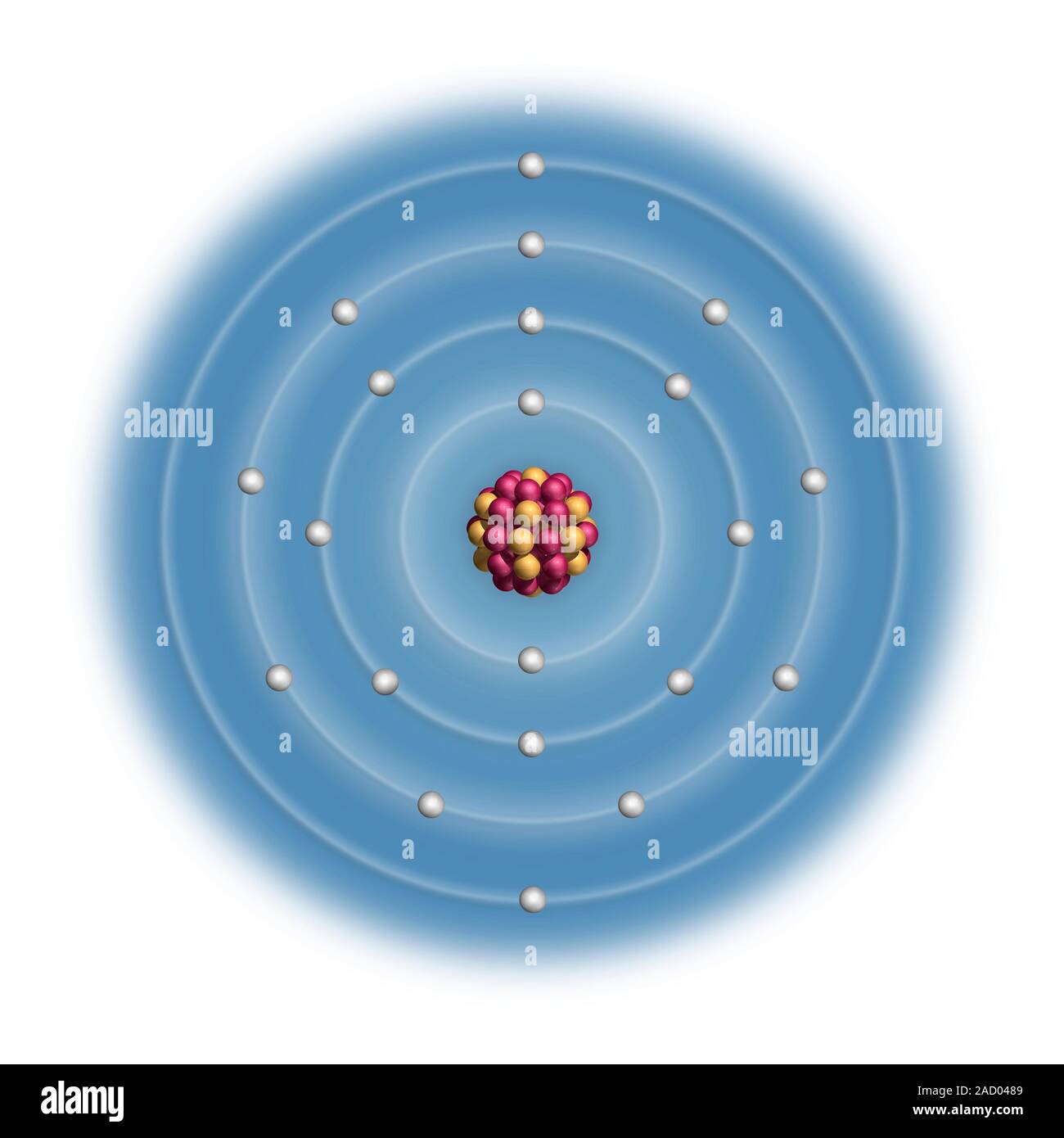
Even though its electron configuration is a bit different, scandium still wants to bond with those three extra electrons, no matter what shell they are in. You will find scandium to the right of calcium in the fourth period of the table. Instead of having three electrons in the outer shell, scandium adds its electron to the second to last shell. Every element adds one more electron to the outermost shell. Up to this point, you have been using a system of 2-8-8 to describe electron shells. Scandium is the first element of the period table where electron configurations start getting a little wild. The fourth row of the periodic table has transition metals ranging from scandium (21) to zinc (30).Īhh, the transition metals. Remember that the first eight were placed during our trip through the third period/row. When scandium forms its 3+ ion, it loses the 4s2 and the 3d1 electrons to have the. For the fourth period/row, all of these electrons build the third shell to a maximum of 18 electrons. The electronic configuration of Scandium 1s2, 2s2 2p6, 3s2, 3p6, 3d1, 4s2. The ground state electron configuration of ground state gaseous neutral scandium is Ar. 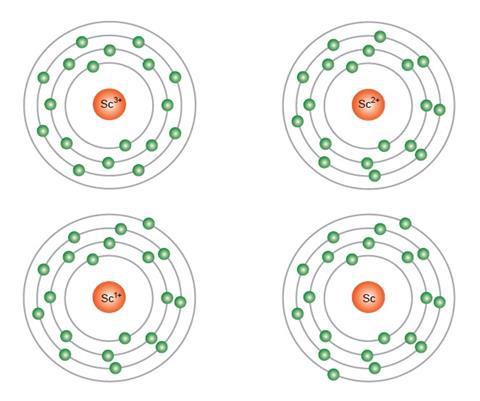
This element is one of the transition elements that doesn't place the additional electrons in the outer shell, but in the one underneath. Scandium atoms have 21 electrons and the shell structure is 2.8.9.2. So remember when you look at our breakdown that the electrons aren't always in a nice neat order as shown here. 
They are found in clouds that can have different shapes that include spheres and dumbbell-like shapes. But conventional wisdom claims that the final. Electron configurations of the elements (data page) 1 H hydrogen : 1s 1s 1 2 He helium : 1s 1s 2 3 Li lithium : He 2s 1s 2s 2, 1 4 Be beryllium. 
As you learn more about atomic structure, you will learn that the electrons don't stay in defined areas around the nucleus. According to the aufbau diagram the configuration should be 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d1 and indeed it is. The electrons like to be in separate shells/orbitals. In an atom, the electrons spin around the center, also called the nucleus. Each of those colored balls is an electron. If you think this is a little over your head, go back and look at the elements 1-18 that have organizations that are a little more simple. Thallium has two possible cationic forms, +1 and +3. They have electron configurations that are made up. The period four transition metal elements have the simplest electron configurations of all the different types of transition metal elements. Beginning with hydrogen, and continuing across the periods of the periodic table, we add one proton at a time to the nucleus and one electron to the proper subshell until we have described the electron configurations of all the elements. Thallium was used as a poison in the Agatha Christie mystery story The Pale Horse. The electron configuration of an element is a series of baseline and superscript symbols that describes the distribution of electrons in atomic orbitals. Let's take a look at the arrangements of electrons in the basic elements (left and right sides of the table) of period four and the more complex arrangements of the transition elements (in the middle of the row). To determine the electron configuration for any particular atom, we can build the structures in the order of atomic numbers. You may have an easy way to know the number of electrons in a neutral atom, but the placement of those electrons gets a little more complex. Now we're working with the fourth period/row in the table of elements. The IB accepts either Ar4s 2 3d 1 or Ar3d 1 4s 2 as the correct electron configuration for scandium. It tells you the mass of one atom, how many pieces are inside, and where it should be placed on the periodic table. In most periodic tables, lanthanum and actinium are. That box on the left has all of the information you need to know about one element. The Group 3B elements (Group 3 in the IUPAC designation) usually have electron configuration (n-1)d1 ns2. The Shorthand electron configuration (or Noble gas configuration) as well as Full electron configuration is also mentioned in the table. Electron configuration chart of all Elements is mentioned in the table below. Complete ground state electronic configuration for the.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
